Today, Brain Windows welcomes its first guest contributor. Dr. Jasper Akerboom is a post-doctoral associate in the lab of Loren Looger at Janelia Farm, and is the lead author on a recently published report on the structure of the genetically-encoded calcium sensor, G-CaMP2. We are very grateful for his contribution!
After the previous post describing G-CaMP2 crystallization two papers describing the crystallization and structure determination appeared online:
Crystal structures of the GCaMP calcium sensor reveal the mechanism of fluorescence signal change and aid rational design. Akerboom J, Vélez Rivera JD, Rodríguez Guilbe MM, Alfaro Malavé EC, Hernandez HH, Tian L, Hires SA, Marvin JS, Looger LL, Schreiter ER. J Biol Chem. 2008 Dec 18.
and
Structural Basis for Calcium Sensing by GCaMP2. Wang Q, Shui B, Kotlikoff MI, Sondermann H.Structure. 2008 Dec 12;16(12):1817-27.
Both papers are very similar, with minor differences in the approach of some of the problems, which will be described below.
In the paper of Wang et al., crystallization of GCaMP2 is achieved by removing the pRSET tag, important for in vivo GCaMP2 function (Nakai et al). Removal of disordered expression tags often is essential for protein crystallization, however, Akerboom et al crystallized GCaMP2 with this tag still present. Spectrophotometric properties of purified GCaMP2 protein with and without pRSET module are identical.
Both in the JBC paper as well as the Structure paper the authors describe the presence of dimeric calcium loaded GCaMP2, appearing as a minor fraction during gel filtration analysis.

Size-exclusion trace of calcium loaded (blue line) and calcium free (red line) G-CaMP2
Akerboom et al initially only crystallized the dimeric form of GCaMP2. Attempts to crystallize monomeric GCaMP2 failed. Selection and mutagenesis of amino acids partaking in the dimer interface in GCaMP2 resulted in the subsequent crystallization of monomeric GCaMP2. Wang and coworkers were able to crystallize both forms without mutagenesis, although their GCAMP2 molecule had its pRSET module removed, indicating a potential role for the pRSET peptide in dimerization.
Both monomeric and the dimeric crystal forms described in both papers are essentially the same.
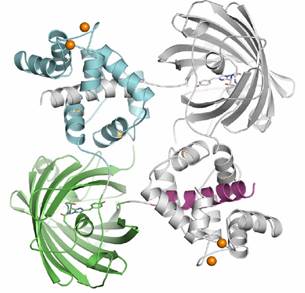
Dimeric G-CaMP2

Monomeric G-CaMP2
The dimeric form of G-CaMP2 is a domain swapped dimer with the M13 peptide (magenta) of each monomer bound by the calcium loaded CaM domain (cyan) of the other. The monomer is very different from the dimer, with the M13 peptide bound by the CaM domain of the own molecule. The interface between CaM and cpEGFP is considerably different between the two different oligomeric states of G-CaMP2.
Wang hypothesizes about a potential role of residue T116 (T203 in GFP numbering) playing in chromophore stabilization in calcium saturated G-CaMP2; this residue adopt a different rotamer in the dimeric structure, in a way that this threonine cannot partake in the hydrogen bond network, dimeric G-CaMP2 is less bright. In the paper by akerboom et al this residue adopts double conformations, so its not clear if this residue is actually the reason for this effect. In addition the mutation T203V results in increased fluorescence in G-CaMP2. Valine is hydrophobic and cannot participate in hydrogen bond formation at all.
Both groups performed mutational analysis of G-CaMP2. Both groups actually described a few identical positions (R81 and R377), and came roughly to the same conclusions, R81 and R377 play a role in the calcium loaded state of the protein. Wang et al performed the experiment using both mutations, and showed a profound decrease of fluorescence.
The group from Janelia Farm made some efforts to improve sensor functionality, and showed that replacing an aspartate close to the chromophore in the calcium saturated state with a tyrosine increases fluorescence by lowering the percentage of protonated chromophore.
Both Wang et al and Akerboom et al tried to study apo-G-CaMP2. Wang and co-workers used small-angle X-ray scattering (SAXS) of apo-G-CaMP2 and solved the structure of cpEGFP. The other group mutagenised all four EF-hands of CaM, removing the calcium binding capacity of G-CaMP2, and subsequently crystallizated the calcium binding deficient G-CaMP2. Both SAXS and crystallization indicated a more open structure of GCaMP2 compared to the calcium loaded state.


SAXS with fitted cpEGFP and 3CLN structures

apo G-CaMP2 structure
In the crystal structure, the M13 peptide and the C-terminal domain of CaM are disordered, indicating the large degree of freedom in apo G-CaMP2. Part of the linker between the M13 peptide and cpEGFP in the apo structure forms part of the beta barrel of cpEGFP.
Both papers will contribute to the understanding of the GECI G-CaMP2. Further directed mutagenesis studies on the basis of the results described in both manuscripts will hopefully result in a better sensor for in vivo imaging.
Leave a comment